How FDA and NYDH Licensing Ensure the Highest Standards in Fertility Care
Navigating fertility treatment options can feel overwhelming, especially when considering treatment abroad. However, understanding the regulatory standards that protect patient safety is crucial for making informed decisions about your fertility journey, which can give you peace of mind to move forward with this exciting process! FDA and NYDH licensing provide essential safety frameworks, ensuring the highest quality care for fertility treatment patients. We are proud to abide by these guidelines when working with intended parents as we help them build a family of their own.
At Chedid Grieco, we prioritize patient safety through our strict adherence to both FDA and NYDH licensing requirements. As one of only eight clinics outside the U.S. with a New York tissue license, we combine Brazilian reproductive expertise with rigorous American safety standards to provide exceptional care for our international patients seeking to build their families.

Understanding FDA Oversight in Fertility Care
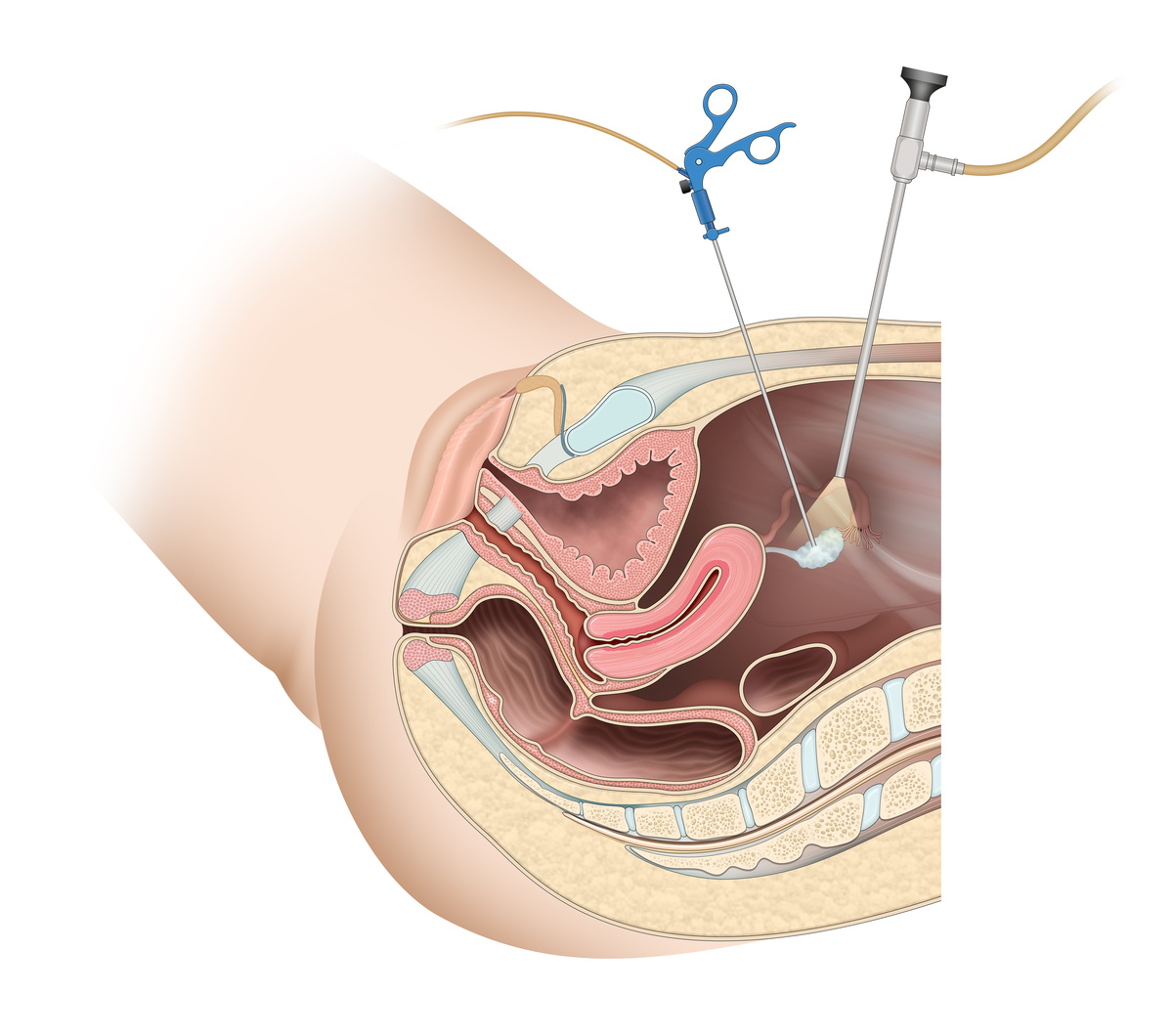
Many intended parents are surprised to learn that the U.S. Food and Drug Administration (FDA) is critical in regulating reproductive tissues like eggs, sperm, and embryos. FDA oversight focuses primarily on preventing disease transmission and ensuring the safety of these biological materials in fertility treatments. This regulatory framework requires licensed facilities to implement comprehensive screening protocols for infectious diseases.
When a fertility clinic obtains FDA licensing, it must follow strict guidelines for donor screening, testing, and record-keeping. According to the Centers for Disease Control and Prevention, proper screening protocols can dramatically reduce the risk of disease transmission through reproductive tissues when intended parents undergo certain fertility treatments like Invitro Fertilization or egg donation. These regulations help protect both intended parents and any children born through fertility treatments.
FDA licensing provides crucial peace of mind for intended parents who work with international clinics like Chedid Grieco. When working with our FDA-licensed facility, you can trust that all reproductive tissues meet the same safety standards required within the United States. This regulatory oversight includes the following:
- Thorough medical and genetic screening of donors
- Comprehensive infectious disease testing
- Careful documentation of all procedures
The FDA also monitors tissue handling procedures to prevent contamination or mix-ups. Licensed facilities must maintain detailed records of every step in the process, from collection to storage to transfer, creating a transparent chain of custody for all reproductive tissues.
The Significance of New York Department of Health Licensing
The New York Department of Health (NYDH) tissue licensing represents one of the most rigorous regulatory standards in reproductive medicine worldwide. New York State maintains exceptionally high requirements for facilities handling reproductive tissues, often exceeding federal guidelines in their thoroughness and scope. NYDH tissue licensing covers every aspect of reproductive tissue handling, including:
- Laboratory protocols and quality control measures
- Staff qualifications and ongoing training requirements
- Facility design and maintenance standards
- Emergency preparedness procedures
- Patient consent processes and documentation
Having NYDH licensing means our fertility clinic meets or exceeds these demanding standards, even when operating outside the United States. This credential is particularly valuable for intended parents who want assurance that their care abroad will match or exceed what they would receive at home in the United States.
The comprehensive nature of NYDH licensing extends to ethical considerations as well. Licensed facilities must adhere to strict informed consent requirements, ensuring patients fully understand all aspects of their treatment. These regulations also mandate detailed record-keeping that protects patients’ rights to information about their care, ensuring that intended parents know every step of their journey!
How Dual Licensing Benefits Fertility Patients
When a fertility clinic maintains both FDA and NYDH licensing, patients receive the benefits of dual regulatory oversight. This combination creates a robust safety framework that addresses both biological safety concerns and operational quality standards.
The dual licensing approach is particularly valuable in fertility tourism, where patients may worry about variable standards across different countries. By choosing a clinic with both FDA and NYDH licensing, you can be confident that your care meets recognized international benchmarks for safety and quality, regardless of where treatment occurs.
These licensing credentials also facilitate better coordination between international and U.S.-based healthcare providers. Because licensed facilities maintain documentation that meets American standards, it’s easier for your home physicians to understand and support your treatment plan. This coordination becomes especially important for any follow-up care needed after returning home. Dual licensing often indicates a clinic’s commitment to continuous quality improvement. Maintaining these credentials requires regular inspections and updates to protocols based on evolving best practices in reproductive medicine. This ongoing oversight helps ensure that patients receive the most current, evidence-based care available when undergoing fertility care in hopes of welcoming a child into their lives.
Ready to Experience World-Class Fertility Care? Contact Chedid Grieco Today!
If you’re considering fertility tourism as part of your family-building journey, Chedid Grieco offers an exceptional combination of Brazilian reproductive expertise with rigorous American safety standards. As a female-led company with 30 years of experience and nearly 9,000 babies born through our care, we understand both the emotional and medical complexities of fertility treatment. Our status as one of only eight clinics outside the U.S. with a New York tissue license reflects our unwavering commitment to patient safety and care quality.
Don’t let geographical boundaries limit your fertility options. Our team provides personalized consultations in Miami while conducting treatments at our state-of-the-art facility in Brazil. There, we offer innovative techniques like Invitro Maturation, which can eliminate the need for ovarian stimulation injections. Experience the boutique care and customized treatment plans that have made us the Head of the Brazilian Technique in reproductive medicine. Contact us today at (305) 912-0050 or through our contact form to begin your journey toward building the family you’ve always wanted.